Fresh Starts, Real Readiness: Turning Site Initiation into a Trial Success Multiplier
By leveraging the Fresh Start Effect at site initiation and shifting from passive training to demonstrated readiness, sponsors and CROs can turn trial launch into a powerful multiplier for early performance, compliance, and execution quality.
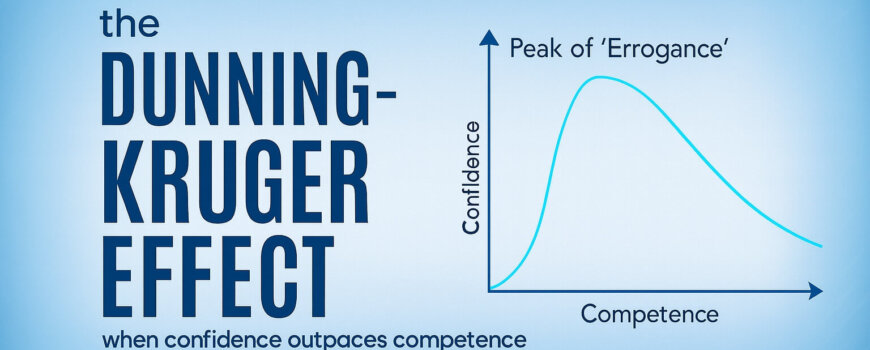
The Confidence Trap in Clinical Trials: When Knowing Just Enough Becomes Dangerous
When confidence outpaces competence in clinical research, the risks can be hidden…and costly for trial sponsors
Running Smarter Trials When Every Dollar Counts
Clinical trials aren’t just more complex. They’re harder to manage, more expensive to run, and increasingly exposed to delay and disruption. Sponsors willing to take a different path can deliver faster, higher-quality trials at a lower cost.
Reflections from DIA and BIO: What Trial Leaders Are Really Saying
What We Think We Know: How Overconfidence Derails Clinical Trials
Why Mindset Matters in Clinical Research
Clinical trials professionals face a host of challenges: for some, it’s the complexity in protocol design, for many it’s lagging enrollment rates, and for others it’s the site burden that’s inherent to change management.
The Compounding Power of Training: Early Investment Yields Exponential Returns
The compounding benefits of effective training cannot be overstated. When clinical research associates (CRAs) and site staff are thoroughly prepared, they engage more deeply with the trial protocols, ask more insightful questions, and more proactively anticipate challenges.
Embracing Change and Intentionally Living ‘a Life of Why
I was recently invited to deliver a keynote presentation on embracing change. The audience was more than a 100 healthcare improvement and clinical trial professionals who had each committed to participate in a year-long collaborative.
Raising the Bar: Why Quality and Training Will Define Your Success Under ICH E6(R3)
The formal Formal adoption of the E6(R3) guideline introduces a proportionate and more modern risk-based approach to quality management. Clinical trial leaders must implement more effective training programs that address increased trial complexity and ensure compliance with the enhanced quality requirements.