
Predict and improve clinical trial
site and team performance.
Accelerate timelines, mitigate risk, and reduce burden
Introducing Ready
A Smarter Way to Accelerate Enrollment and Minimize Risks
Ready by ArcheMedX transforms study startup to prevent issues downstream by delivering better training that predicts and improves site and study team performance.
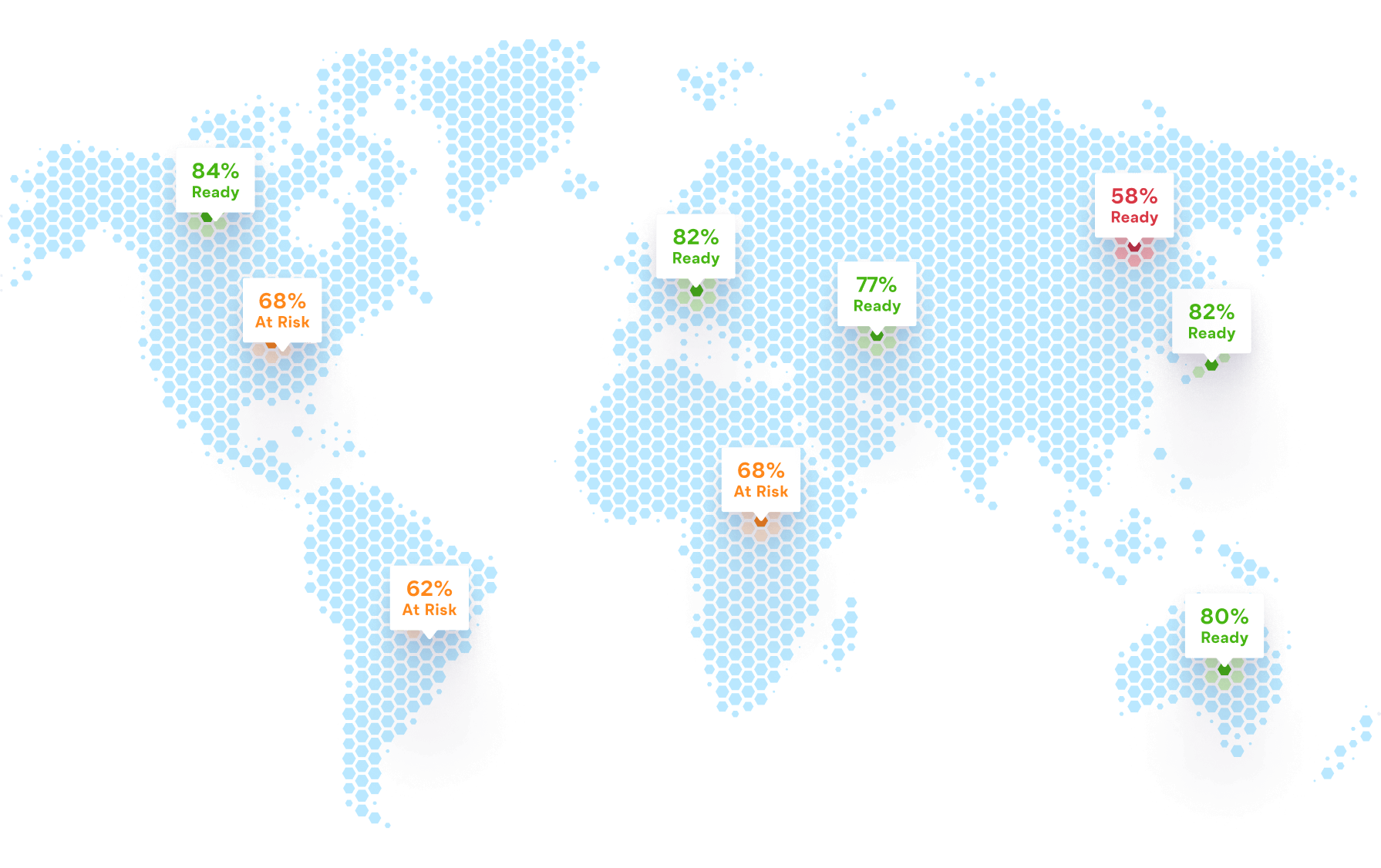
Ready streamlines site initiation and reveals which teams and sites
are best prepared to successfully conduct your study
Optimize Study Team Training
Save time and money while ensuring sites and CROs are well prepared and compliant
Identify Sites Ready to Screen and Enroll
Accelerate timelines and avoid preventable delays and protocol deviations
Inform Risk Based Quality Management
Optimize resource deployments, minimize downstream risks, and improve quality
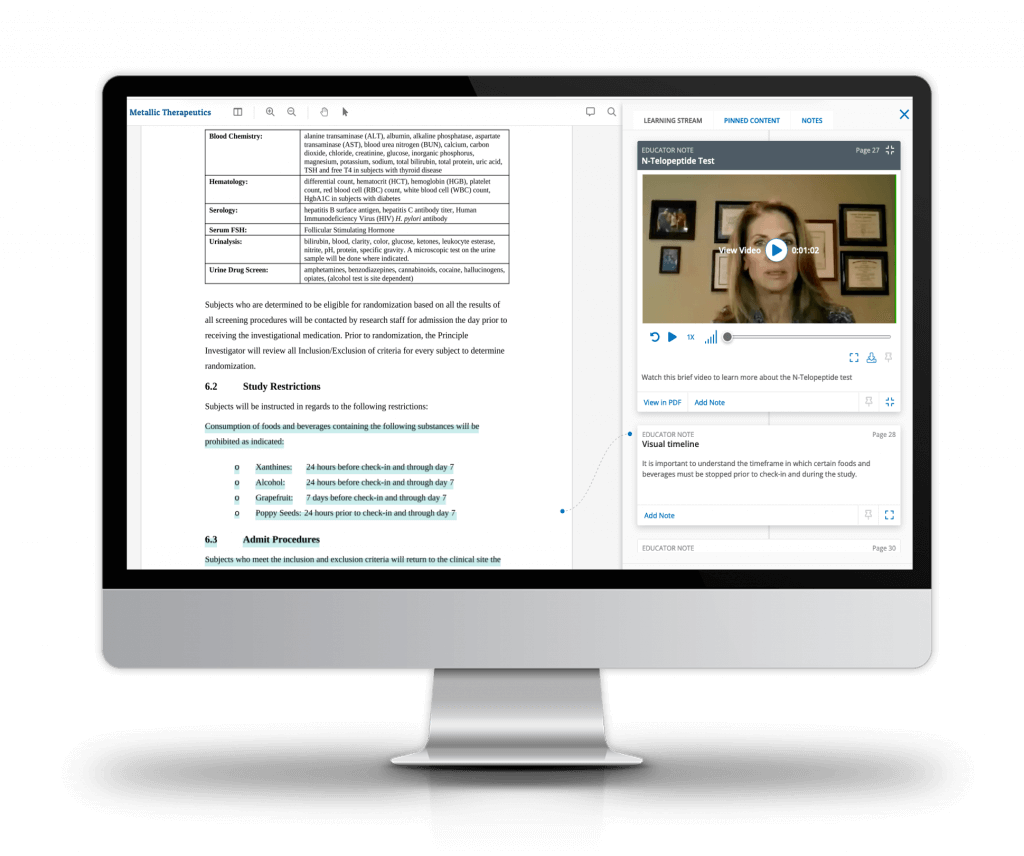
Ready delivers more effective study training
Ready’s behavioral science-based approach enhances role specific training and improves performance:
- Eliminate clumsy slide presentations that miss critical information
- Rapidly convert study documents into interactive learning experiences
- Deliver training on-demand; tailored to each role, site, or geography
- Automate training assignments and personalized learning paths
- Tailor remediation based on identified training gaps
Allow individuals to train on-demand across any device without the burden of additional passwords or downloading another app
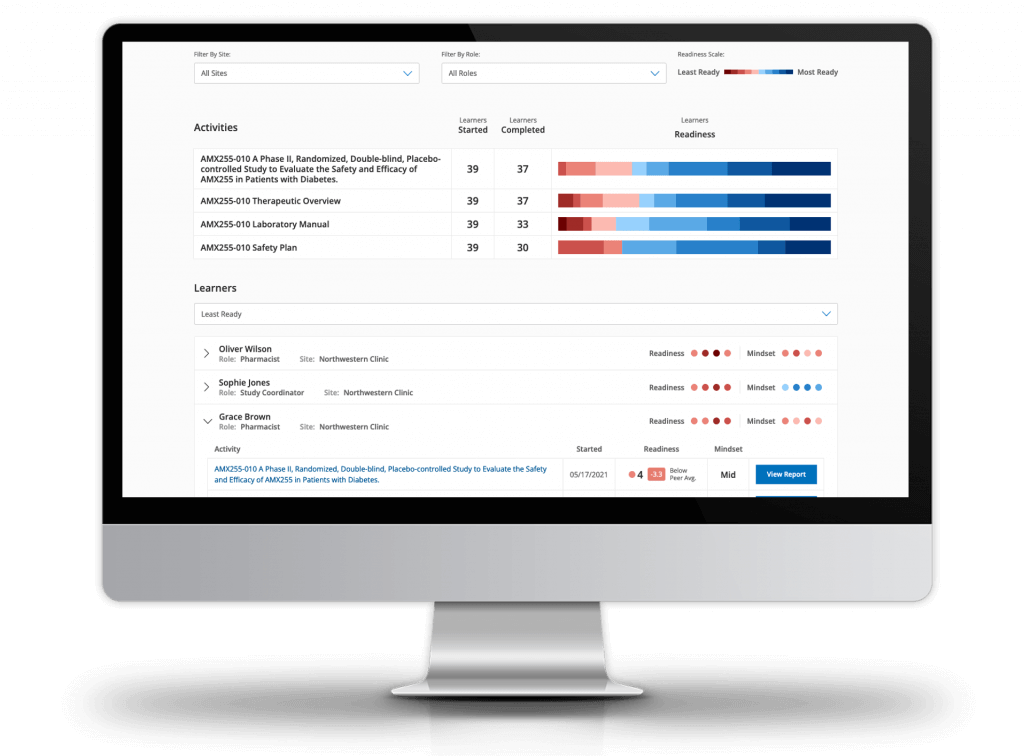
Ready measures study knowledge and predicts trial performance
Ready’s unique behavioral analysis model identifies top performers and reveals areas of study risk sooner:
- Access dashboards to track progress, identify risks, and predict performance
- Confirm each site’s knowledge and confidence to conduct the study
- Proactively identify and addresses areas of confusion in study documents
- Inform faster operational decisions and RBM strategies
- Digitize training certificates and seamlessly file in your eTMF
90% SIV Reduction
96% Screen & Enroll Sooner
ArcheMedX is reducing protocol deviations, accelerating enrollment,
and reducing site/CRO burden.
Emerging Biotech Streamlines Site Initiation
By deploying Ready, an emerging biotech and mid-sized CRO re-imagined site initiation to convert the traditional PI meeting and SIVs into a tailored, on-demand training experience powered by Ready. As a result, the trial sponsor streamlined the overall initiation process and reduced their SIVs from 4+ hours to 30 minutes.
Global CRO Transforms Clinical Delivery
A top five CRO dramatically improved its workforce performance management utilizing Ready to change the way clinical delivery managers and directors evaluated and equipped their project teams to more rapidly identify, upskill, and deploy the most capable staff on global clinical studies.
Research Center Reduces Staff Evaluation Time by 40%
A nationally recognized academic research center utilized Ready to accelerate on-boarding for new project members and accurately predict high and low performers. The program manager reported to leadership that using Ready would streamline staff evaluations, increase preparedness, and ultimately reduce time by 40-50%.
